Poly(D, L-lactide)’╝īPDLLA

Racemic polylactic acid (Poly(D,L-lactide), PDLLA) is an amorphous polymer composed of both left-handed (L-type) and right-handed (D-type) isomers, possessing unique physical and chemical properties, as well as excellent biocompatibility and degradability.
PDLLA has a wide range of applications in the medical device field. It can be used as facial filler material in medical aesthetics, drug-eluting coatings, drug-loaded microspheres or particles for sustained drug release carriers; bone fixation devices or porous scaffolds for tissue engineering; implants and alveolar bone repair; postoperative anti-adhesion films; surgical sutures; hemostatic clips; retinal ophthalmic implant materials, and more.
┬Ā
eSUNMED PDLLA Material Features
White granules, non-irritating, amorphous polymer, non-toxic, biodegradable and absorbable, easy to process and mold, with a degradation period typically ranging from 3 to 24 months.
Performance Indicators of eSUNMED’s Implant Grade PDLLA Material
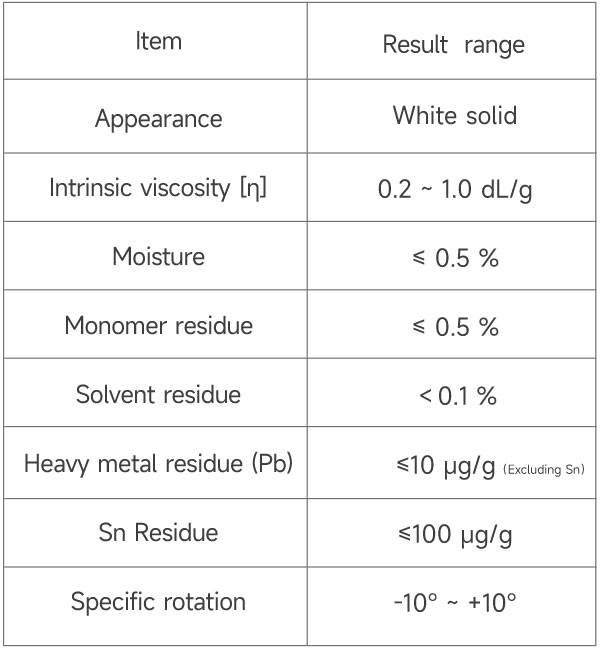
*┬ĀeSUNMED can provide medical raw materials with different molecular weights or characteristic viscosities according to customer requirements.
Performance Indicators of eSUNMED’s Implant Grade PDLLA Microspheres
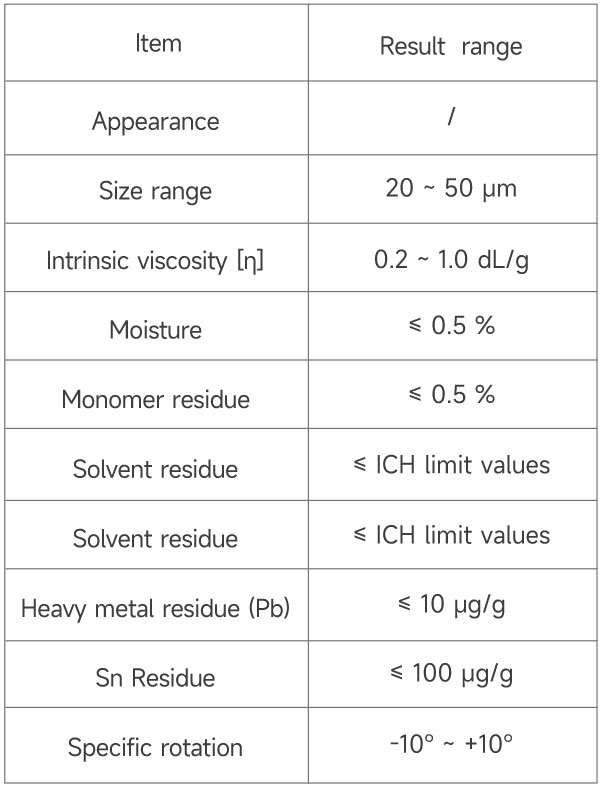
*┬ĀOn June 8, 2024, eSUNMED medical PLLA microspheres completed the main document registration and filing.
Common Applications of PDLLA Material
1.┬ĀFacial Filler Material
PDLLA material is prepared into microspheres of 20~50╬╝m, serving as an excellent subcutaneous stimulatory filler. It shares similar characteristics with PLLA microspheres, but due to PDLLA being an amorphous polymer and non-crystalline, it degrades faster both inside and outside the body. There are also differences in the internal and external morphology of PDLLA microspheres compared to PLLA microspheres. Overall, PDLLA microspheres, as a new type of facial filler material, exhibit excellent biocompatibility, biodegradability, and the ability to stimulate collagen regeneration upon degradation. Currently, medical aesthetic filler products based on PDLLA microspheres have been widely used in clinical applications.
Approved for the market, there are already AestheFill PDLLA microsphere facial fillers available, and eSUNMED can provide PDLLA raw materials and microspheres for matching filler products.
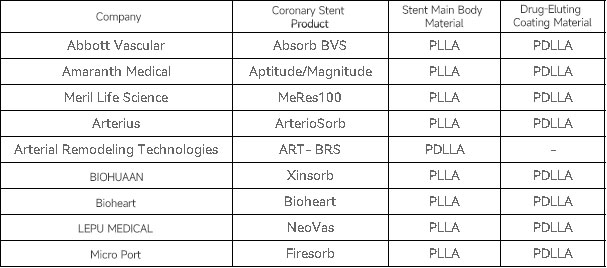
2.┬ĀPDLLA Drug-Eluting Coatings
PDLLA drug-eluting coatings are extensively used in degradable coronary stents. Representative product application examples are as follows:
3.┬ĀBone Repair and Tissue Engineering Scaffolds
The glass transition temperature of PDLLA is approximately 50-60┬░C, which makes it suitable for printing. It can be easily printed into porous scaffolds, as shown in the stereolithography-constructed PDLLA tissue engineering scaffold below. [3] Its elastic modulus is comparable to that of cancellous bone, which is beneficial for bone healing, and it has good degradability and biocompatibility. However, PDLLA material lacks osteoconductivity and has a slow bone defect repair rate. It is often blended with inorganic materials that have good tissue compatibility, osteoconductivity, and bioactivity to prepare composite materials. Numerous studies have reported the application of PDLLA materials in bone repair and tissue engineering scaffolds. Some absorbable bone fixation devices and maxillofacial bone repair instruments have been successfully applied clinically, showing good tissue repair effects and wide applications in the field of tissue engineering.

Image: Stereolithography-constructed PDLLA tissue engineering scaffold. (A) Optical photograph, (B) micro-CT visualization, (C) SEM image, (D) Light microscope image of the scaffold after 1 day of osteoblast culture in mice. The scale represents 500 micrometers. [3]
4.┬ĀOther Application Areas
PDLLA material has been extensively researched and applied in dental implants, alveolar bone, and periodontal repair; PDLLA anti-adhesion films have been used for decades in clinical settings to prevent postoperative adhesions in conditions such as herniated discs, gallstones, appendicitis, and uterine tumors; moreover, PDLLA has a wide range of applications in absorbable surgical sutures, hemostatic clips, ophthalmic implants, and other areas.
About eSUNMED
eSUNMed Biotechnology (Shenzhen)Co., Ltd. (brand “eSUNMED”) is primarily dedicated to the research and industrialization of biomedical polymer materials, medical 3D printing materials, and downstream medical supplies and equipment.
Currently, eSUNMED can provide customers with medical-grade raw materials of various molecular weights such as PLA and PCL. Additionally, based on customer needs, it can customize solid microspheres of the aforementioned polymers with diameters ranging from 10 to 100╬╝m.
eSUNMED is located in Longhua District, Shenzhen, with an operational base covering more than 2000 square meters. It has a 100,000-level standard clean workshop of nearly 400 square meters and a 10,000-level purification laboratory of 100 square meters, meeting the standards for auxiliary drug material production and sterile medical device production in GMP-compliant factories, in line with the industry standards for biomedical polymer materials.
Note: Parts of this article are referenced from the following literature:
[1] Jinnouchi H, Torii S, Sakamoto A, et al. Fully bioresorbable vascular scaffolds: lessons learned and future directions[J]. Nature Reviews Cardiology, 2019, 16(5): 286-304.
[2] Lin C Y, Lin J Y, Yang D Y, et al. Efficacy and safety of poly-D, L-lactic acid microspheres as subdermal fillers in animals. Plast Aesthet Res, 2019, 6: 16.
[3] Melchels F P W, Feijen J, Grijpma D W. A poly (D, L-lactide) resin for the preparation of tissue engineering scaffolds by stereolithography. Biomaterials, 2009, 30(23-24): 3801-3809.

